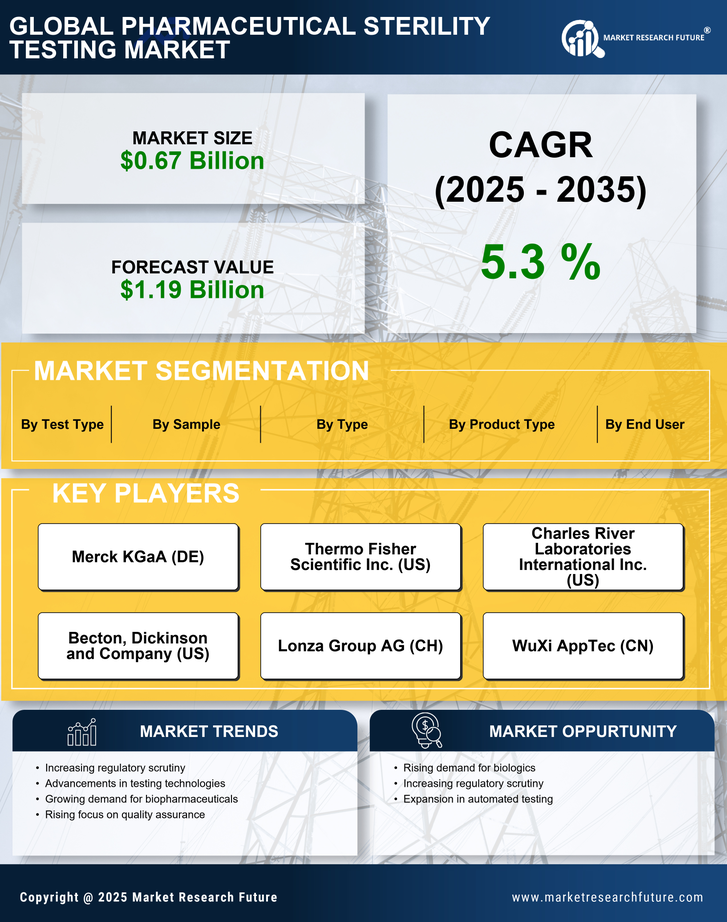
Sterility Testing Supporting Global Vaccine Production
The Pharmaceutical Sterility Testing Market has gained prominence alongside expanding vaccine production efforts worldwide. Vaccines require strict aseptic manufacturing conditions because contamination risks can compromise both safety and effectiveness.
Large-scale immunization programs demand consistent quality assurance throughout production cycles. Sterility testing verifies that each batch meets microbial safety requirements before distribution. This process protects public health and reinforces trust in vaccination programs.
Modern vaccine manufacturing facilities rely heavily on environmental monitoring alongside sterility testing. Air quality, surfaces, and equipment undergo routine microbial assessments to prevent contamination events. Laboratories play a central role in analyzing these samples.
Ref - https://www.marketresearchfuture.com/reports/pharmaceutical-sterility-testing-market-10720
The Pharmaceutical Sterility Testing Market has gained prominence alongside expanding vaccine production efforts worldwide. Vaccines require strict aseptic manufacturing conditions because contamination risks can compromise both safety and effectiveness.
Large-scale immunization programs demand consistent quality assurance throughout production cycles. Sterility testing verifies that each batch meets microbial safety requirements before distribution. This process protects public health and reinforces trust in vaccination programs.
Modern vaccine manufacturing facilities rely heavily on environmental monitoring alongside sterility testing. Air quality, surfaces, and equipment undergo routine microbial assessments to prevent contamination events. Laboratories play a central role in analyzing these samples.
Ref - https://www.marketresearchfuture.com/reports/pharmaceutical-sterility-testing-market-10720
Sterility Testing Supporting Global Vaccine Production
The Pharmaceutical Sterility Testing Market has gained prominence alongside expanding vaccine production efforts worldwide. Vaccines require strict aseptic manufacturing conditions because contamination risks can compromise both safety and effectiveness.
Large-scale immunization programs demand consistent quality assurance throughout production cycles. Sterility testing verifies that each batch meets microbial safety requirements before distribution. This process protects public health and reinforces trust in vaccination programs.
Modern vaccine manufacturing facilities rely heavily on environmental monitoring alongside sterility testing. Air quality, surfaces, and equipment undergo routine microbial assessments to prevent contamination events. Laboratories play a central role in analyzing these samples.
Ref - https://www.marketresearchfuture.com/reports/pharmaceutical-sterility-testing-market-10720
0 Commentarios
0 Acciones
87 Views
0 Vista previa